
Conference & Exhibition Venue
Congress Center Basel
Messeplatz 21

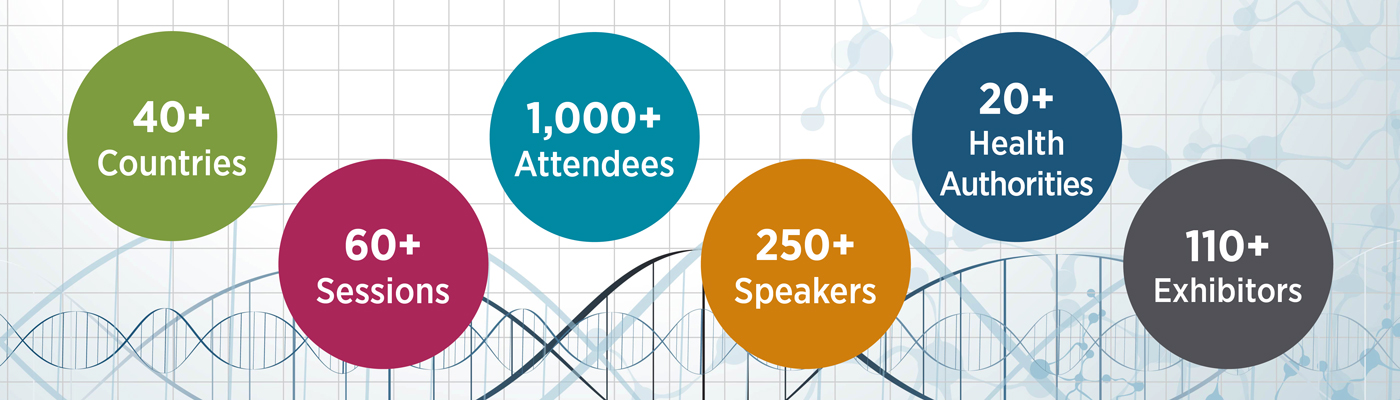
Welcome to DIA Europe
2018 marks the 30th anniversary of the DIA EuroMeeting. To support this anniversary, and to align this European flagship meeting with all the DIA regional flagship meetings occurring across the globe, we are renaming the gathering, DIA Europe 2018. This new name reflects not only regional alignment but also a change in approach to more directly and proactively focus on the expressed needs and interests of our constituents.
DIA Europe 2018 is more than just a traditional meeting. It’s a chance to learn, engage, and grow as a professional in health care.
We convene these platforms around the globe, from the DIA Global Annual Meeting in the United States, to regional meetings in China, Europe, India and Japan, to provide you with a neutral forum to discuss current issues, with the goal of uncovering new ways of working and new solutions for patients. We welcome participants from across the entire drug development industry. By incorporating representatives from the full life sciences landscape we facilitate open collaboration. We ensure that industry, payors, patients and regulators, all have an equal voice.
We look forward to welcoming everyone to DIA Europe's home in Basel, Switzerland.